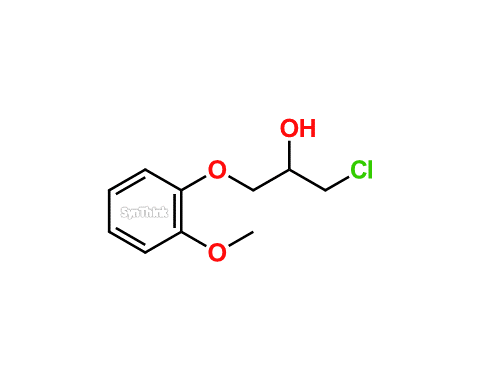
SOLVED: The heat of combustion of propan-2-ol at 298.15 K, determined in a bomb calorimeter, is -33.41 kJ / g . For the combustion of one mole of propan-2-ol, determine (a) Δ

Circle K Tjøme - Fortvil ikke :-) Når du kjøper propan på flaske fra oss er du sikret en godkjent flaske til enhver tid. Det du kjøper er en rettighet i AGAs
![Potassium;1-[4-(3-methylanilino)pyridin-3-yl]sulfonyl-3-propan-2-ylurea | C16H20KN4O3S+ | CID 20025616 - PubChem Potassium;1-[4-(3-methylanilino)pyridin-3-yl]sulfonyl-3-propan-2-ylurea | C16H20KN4O3S+ | CID 20025616 - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=20025616&t=l)
Potassium;1-[4-(3-methylanilino)pyridin-3-yl]sulfonyl-3-propan-2-ylurea | C16H20KN4O3S+ | CID 20025616 - PubChem

Excess volume (top) and excess enthalpy (bottom) of propan-2-ol + water... | Download Scientific Diagram

Circle K Korgen - PROPAN INFO: • Vi bytter kun flasker, det er ikke pant og retur på tomme flasker ifølge AGAs kjøpsbetingelser. • Vi bytter ikke utenlandske flasker, eller noen av



![IR Spectra for 1,2,5-trimethyl-3-[(2R)-2-(propan-2-yl)pentyl]benzene | Mol-Instincts IR Spectra for 1,2,5-trimethyl-3-[(2R)-2-(propan-2-yl)pentyl]benzene | Mol-Instincts](https://static.molinstincts.com/compound_common/1-2-5-trimethyl-3-2R-2-propan-2-yl-pentyl-benzene-2D-structure-CT1002465297.png)