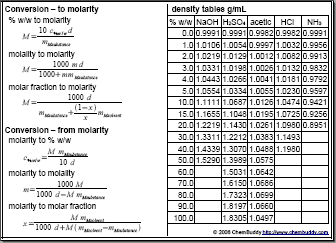
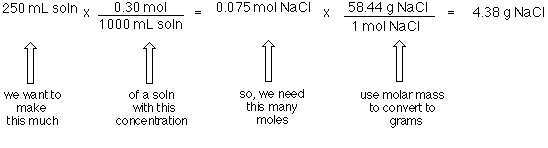SOLVED: Submit Converting molarity to other units of concentration Concentration is typically only expressed using single unit (with an exception being solubility study where another unit of concentration is provided in parentheses
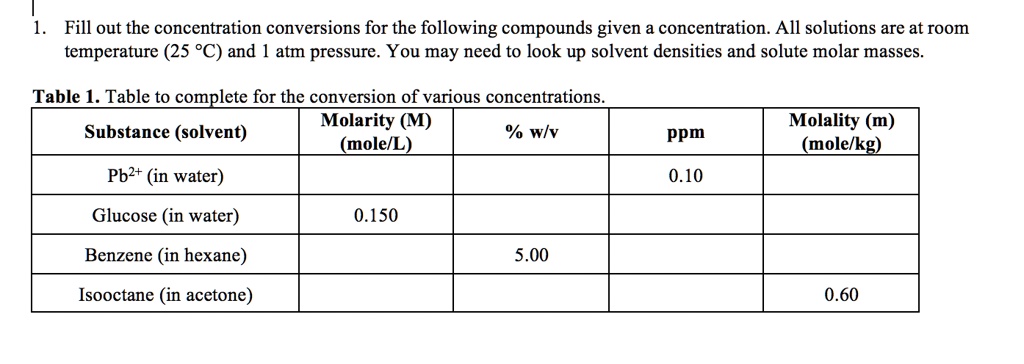
SOLVED: Fill out the concentration conversions for the following compounds given a concentration. All solutions are at room temperature (25 *C) and atm pressure. You may need to look up solvent densities